Science
Neuronal Hyperactivity in various brain regions is associated with anxiety, depression, and other neurocognitive disorders and psychiatric conditions.

Article by Dean Radin, Ph.D., Chairman and Co-founder
in Springer Nature | Resarch Communities
Toward a new class of genetic therapeutics for mental health disorders
Read Article
THE COGNIGENICS APPROACH
Genetic therapeutics represent a transformative advancement in biopharmacology, enabling the precise manipulation of DNA (permanent) or RNA (temporary) sequences. This new class of medicines can insert, delete, or modify specific genetic sequences, allowing for more effective mental health treatments.
Unlike traditional pharma drugs, which are taken systemically and can cause undesirable side effects, genetic medicines offer the promise of targeting only those neuronal receptors or neuronal circuits in the brain that are responsible for the disorder, vastly reducing possible side effects. In addition, unlike genetic-based vaccines, these therapeutic medicines do not introduce foreign proteins into the body, so they do not provoke immune responses, and nor do the RNA treatments affect DNA.
LIMITATIONS OF CURRENT TREATMENTS IN ANXIETY AND DEPRESSION
- Outdated Pharmacological Options
Current anxiolytic and memory drugs were developed in the 1980s and 1990s. This presents a significant opportunity for innovative breakthroughs in treatment approaches. - Limited Effectiveness and Adverse Side Effects of SSRIs
Common SSRIs like Prozac, Celexa, and Zoloft offer limited efficacy and are associated with a range of serious side effects. More effective and safer treatment alternatives are needed. - Inadequate Response Rates to SSRIs
Research indicates that 30% to 50% of patients suffering from anxiety disorders do not find adequate relief with SSRIs. - High Relapse Rates
Individuals with anxiety disorders often face relapse rates between 50% to 80% over several years, even with ongoing treatment. - Adverse Effects of Long-Term Medication Use
Prolonged use of benzodiazepines can lead to drug dependence. Side effects of SSRIs, such as sexual dysfunction, chronic nausea, impaired digestion, dry mouth, weight gain, sleep disturbances, and in extreme cases, increased suicidal ideation, further complicate treatment adherence and effectiveness.
These challenges point to a pressing need for novel, more effective, and safer therapeutic interventions in the realm of mental health.
COGNIGENICS’ BREAKTHROUGH INNOVATION
We have developed a noninvasive delivery platform that overcomes the principal obstacle of transporting genetic edits across the blood-brain barrier. Our approach differs from most contemporary gene-based interventions for neurodegenerative conditions, which involve intravenous delivery, injection into the cerebrospinal fluid, or injection directly into the brain.
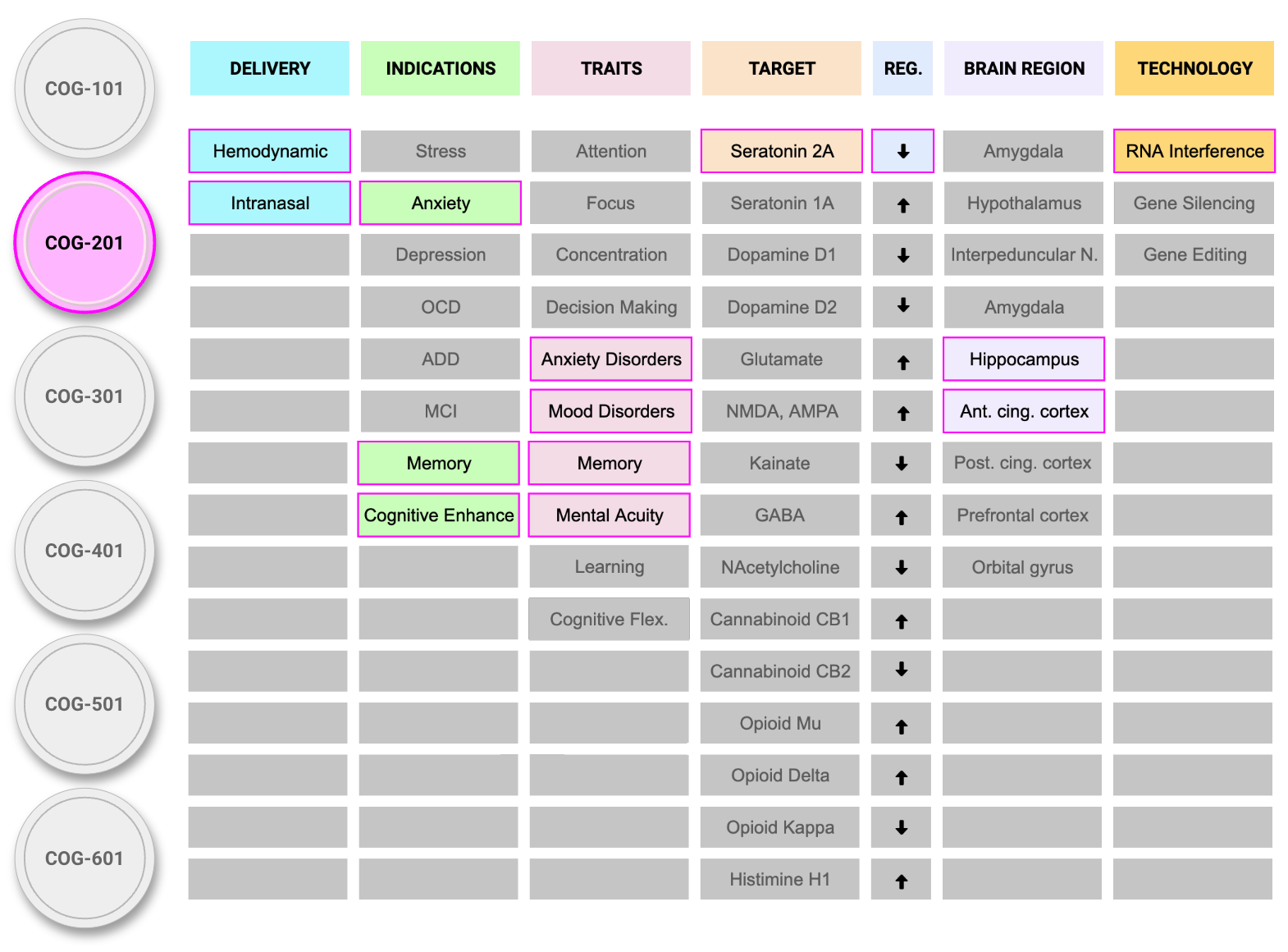
THE COGNIGENICS PLATFORM
Our intranasal delivery platform offers a novel way to provide permanent (via DNA) or long-lasting (via RNA) treatments for mental health disorders associated with hyperactive neurons. The platform can accommodate a wide range of genetic therapeutics, enabling precise treatments that can be customized for individual needs.
ADVANTAGES OF THE COGNIGENICS METHOD
- Enhanced Cost-Efficiency and Accelerated R&D Process
Our strategy involves forming strategic alliances with carefully selected Contract Research Organizations (CROs) for laboratory research. This collaboration not only significantly reduces R&D expenses it also accelerates development. - Accurate Targeting of Neuronal Receptors
Our approach is characterized by its remarkable precision in targeting specific neuronal receptors. This enhances therapeutic efficacy while substantially reducing the likelihood of unintended side effects. - Demonstrated Safety in Preclinical Models
Our preclinical trials using mice and rats have observed no side effects (i.e., no behavioral changes or weight changes). This suggests a high safety profile, which is being investigated in more detail through formal safety and toxicology studies. - Alignment with Current Neuroscientific Foundations
Our methodology approach is based on contemporary findings and hypotheses within the disciplines of neuroscience, neuroanatomy, molecular biology, and genetic engineering.
KEY MILESTONES
- Initiation of Preclinical Research
Our journey began in 2020 with the commencement of comprehensive preclinical studies, laying a solid foundation for our program. - Progress Toward Clinical Trials
We are in the late pre clinical stage of development and are working to enter phase one trials in late 2024. The company will pursue breakthrough designation and accelerated review with a goal of speeding time to market. - Anticipated Market Introduction
We are optimistic about introducing our product to the market by early 2026, provided our R&D continues as planned and we gain FDA approval for clinical trials. This projected timeline is consistent with industry norms.
Our roadmap reflects a blend of innovative research and strategic planning, and is initially aiming to bring a novel and effective therapeutic option to those suffering from chronic anxiety associated with mild cognitive impairment.
